Animal Health and AMR: Livestock Data Analysis
Antibiotics are a cornerstone of modern medicine. However, unnecessary use can lead to ‘resistant’ bacterial infections where antibiotics may no longer work. It is a challenge that cuts across human, environmental and animal health and requires action in all domains. While the animal health sector has worked for many years to support responsible use and manage antimicrobial resistance (AMR), the United Nations acted in 2016 by issuing a ‘Declaration’ signed by 193 countries that committed the world to jointly act.1https://digitallibrary.un.org/record/842813?ln=en This built greater momentum for collective action in animal health, including increasing use of vaccines and diagnostics, improving cooperation across the value chain, and better optimizing antibiotic use.
The result has been:
1) Animals need fewer antibiotics: Use and sales of antibiotics in animals are falling across the world as adoption of preventative tools like vaccines grows, reducing the risk of illness and the need for treatment.
2) Resistance in animals remains low: Monitoring of bacterial infections in animals shows that resistance to critically important antibiotics, in general, remains low and these medicines remain highly effective in animals.
3) Risk of AMR transfer from animals remains low: Animals and food-borne illness are a risk for AMR transfer, but research shows that the primary source of AMR-resistant infections in people remains other people.
4) Protecting people requires a One Health approach: While animal health has taken action, resistance and antibiotic use in humans has risen, which is why a One Health approach is crucial.
Taken together, these results show how responsible use efforts in animals are meeting the objectives of the WHO Global Action Plan on AMR, which focuses on prevention and optimizing use, not simply on reductions.
Animals need fewer antibiotics
Global antimicrobial use in animals is declining
The World Organisation for Animal Health collects global antimicrobial use data and found that “global antimicrobial use in animals has declined by 13% in 3 years,” providing the following figures.2https://www.woah.org/en/new-report-reveals-global-decrease-in-antimicrobial-use-in-animals/ Comparisons with human use are unfortunately not possible as this type of data is not widely collected in human health.

Prevention is reducing the need for antibiotics
Data from the world’s largest animal health companies shows how declines in antimicrobial sales are mirrored by increases in vaccine sales. Prevention reduces the need for treatment.
Global Livestock Vaccine vs Antibiotic Sales, Percent Change (2015 Euros)3Data provided by CEESA. Their International Sales Survey covers sales from the 9 largest global animal health manufacturers (all HealthforAnimals Members) and local/regional manufacturers in Italy, Spain, the UK and Latin America.
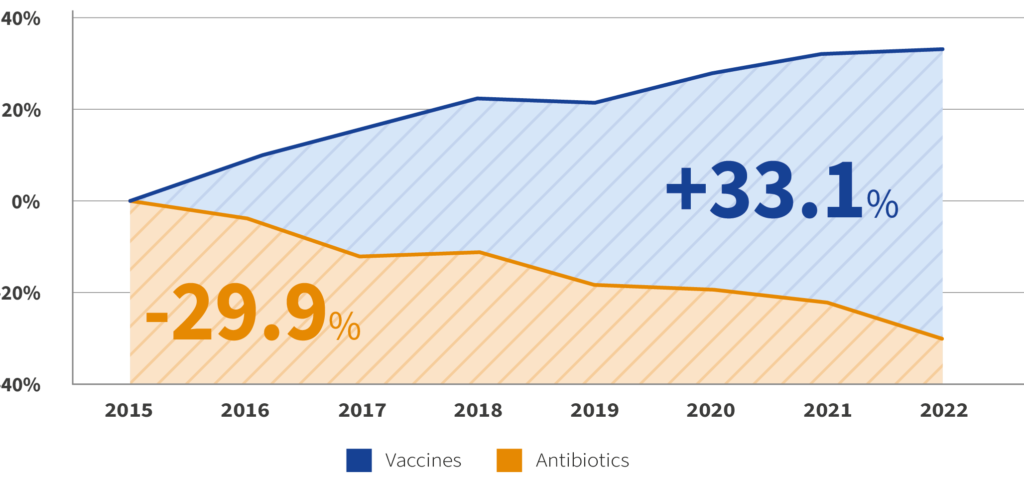
“More progress in agriculture than in the human sector”
Antibiotics consumption in the EU/EEA, 2014–20184Source: Antimicrobial Resistance in the EU/EEA: A One Health Response, https://www.ecdc.europa.eu/sites/default/files/documents/antimicrobial-resistance-policy-brief-2022.pdf
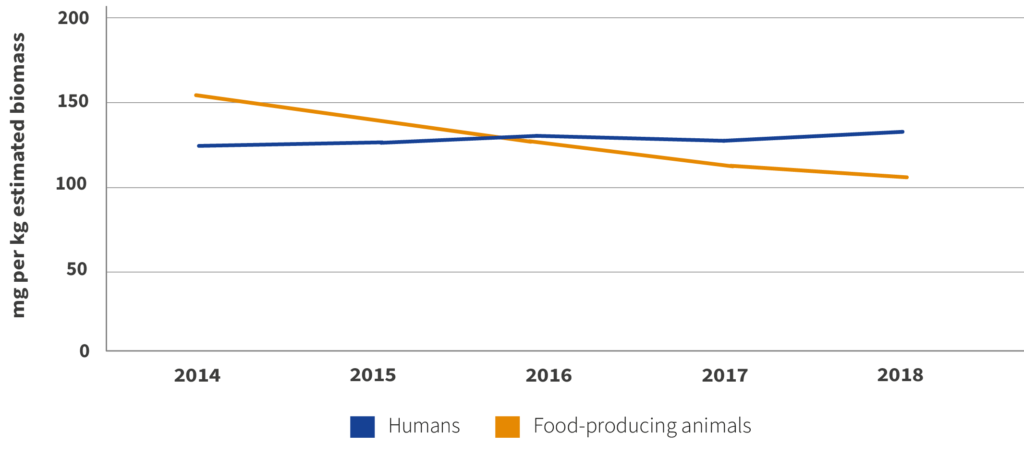
A recent report on antimicrobial use in the EU from the European Medicines Agency (EMA), European Food Safety Authority (EFSA), European Centre for Disease Control (ECDC) and OECD found there was “more progress in agriculture than in the human sector” and since 2016, “average consumption of antibiotics in humans is now higher than in food-producing animals”.5Source: Antimicrobial Resistance in the EU/EEA: A One Health Response, https://www.ecdc.europa.eu/sites/default/files/documents/antimicrobial-resistance-policy-brief-2022.pdf
Antibiotic resistance in animals remains low
Critically important antibiotics are a fraction of overall animal use
The WHO maintains a list of ‘critically important’ antibiotics classes (CIAs) for human health. Some of these are also authorized for use in animals, however these account for “less than 20% of antimicrobials used in animals” according to the World Organisation for Animal Health (WOAH).6https://www.woah.org/en/new-report-reveals-global-decrease-in-antimicrobial-use-in-animals/ Most are also considered ‘Veterinary Critically Important Antibiotics’ by WOAH which means they are “essential” for animal use with no “sufficient alternatives”.
Antibiotic Use in Animals (Critically Important vs Other Classes)7Calculation reflects the forthcoming WHO Medically Important Antimicrobial List where all penicillins are now a ‘highly important’ antimicrobial. Globally: WOAH, 7th Annual Report on Antimicrobial Agents Intended for Use in Animals, USA: USFDA, 2021 Summary Report On Antimicrobials Sold or Distributed for Use in Food-Producing Animals, Europe: 2023 ESVAC Report

Only five percent of animal use is ‘highest-priority’ antibiotics
Comparing human and animal use of critically important antibiotics (CIAs) is challenging. This is because human health relies on WHO’s AWaRe classification for evaluating use, while animal health relies upon the WHO’s Medically Important Antibiotics list. However, the ‘Watch’ category in AWaRe includes “most of the highest-priority critically important antibiotics” (HP-CIAs). This allows for a limited comparison between people and animals focused on these highest priority medicines.

4–5%
of global animal antibiotic use comes from highest-priority critically important antimicrobial classes8Calculation reflects the forthcoming WHO Medically Important Antimicrobial List where Macrolides are no longer an HP-CIA. WOAH’s ‘7th Annual Report on Antimicrobial Agents Intended for Use in Animals’ states that fluoroquinolones and 3rd & 4th gen cephalosporins are 3.4% and 0.6% of animal use respectively. WOAH also stated in direct correspondence that bacitracin is 90% of polypeptide use (8.7%), meaning that only 10% (.87%) could potentially be polymixins, an HP-CIA.

38%
of human use is ‘watch’ antibiotics,938.6% of human use is Watch antibiotics. Reserve is less than 1%. https://www.cidrap.umn.edu/antimicrobial-stewardship/global-use-broad-spectrum-antibiotics-rising-data-show which includes ‘most of the highest-priority critically important antibiotics’10https://adoptaware.org/
Resistance to ‘critically important’ antimicrobials in animals is low
Animal medicines companies and governments regularly monitor resistance to critically important antimicrobials (CIAs) in animals. Their results show that resistance to CIAs remains generally low or absent.

A 14-year review of E. coli in the EU found resistance to critically important antibiotics was generally ‘absent or low’ in animals11https://academic.oup.com/jac/article-abstract/77/12/3301/6750681

In 2021, the UK government found resistance to ‘high-priority’ critically important antibiotics in animals was ‘low or not detected’ for E.coli and ‘no resistance’ or ‘very low’ for salmonella12https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1126449/TP_FINAL_VARSS_Highlights_2021_2022_41102022-accessible.pdf
Risk of AMR Transfer from animals remains low
Most animal antibiotic use comes from just three of the 42 antimicrobial classes available for humans

42
antimicrobial classes are authorized for use in humans, according to the WHO


21
of these classes are also authorized for use in food-producing animals


Just 3
of these 21 classes represent 55% of all animal antibiotic use*
This limited overlap in antibiotic classes means the risk of animals transferring resistant bacteria that would affect the wide range of antibiotic classes available to people is greatly reduced.13List of Antimicrobial classes authorized for humans and animals on page 24: https://cdn.who.int/media/docs/default-source/antimicrobial-resistance/amr-gcp-irc/who_mialist_draft_forexternaldiscussion.pdf?sfvrsn=af6f2ebf_1. Percentages of use in animals can be found in the WOAH Seventh Annual Report on Antimicrobial Agents Intended for Use in Animals
*Penicillins, tetracyclines, and sulfonamides.
Most antibiotic resistant bacteria in people do not come from livestock
Livestock can share antibiotic-resistant bacteria with people through food-borne illness or direct contact with animals. However, studies have found the majority of antibiotic-resistant bacteria are transferred through person-to-person contact, particularly in healthcare settings.
A recent Lancet study analyzed over a decade of data in the Netherlands to identify the source of resistant E. coli genes in people.14https://www.thelancet.com/journals/lanplh/article/PIIS2542-5196(19)30130-5/fulltext They found:


This finding is supported by other studies from around the world:

The UK’s National Institute for Health and Care Research found in another Lancet Study of resistant E. Coli strains – “non-human reservoirs made little contribution to invasive human disease.”15https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(19)30273-7/fulltext

A study in England examined E. Coli strains in both livestock and people, finding that “there was limited evidence that antimicrobial-resistant pathogens associated with serious human infection had originated from livestock.”16https://journals.asm.org/doi/10.1128/mBio.02693-18

Researchers on Reunion Island where “AMR is a major problem” found that “livestock have very little effect on the acquisition of [resistant E.coli] by humans…”17https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9047676/
These studies, when taken together, are why a recent comprehensive literature review18https://pubmed.ncbi.nlm.nih.gov/36136696/ in the Journal of Veterinary Science by researchers across Canada and Europe concluded:
“Most recent studies have reported that livestock have little contribution to the acquisition of AMR bacteria and/or AMR genes by humans”
Livestock have a limited impact on the burden of resistant bacterial disease in people
Animal-sourced foods are considered the primary pathway where antibiotic-resistant bacteria could potentially migrate from livestock to wider human populations. Currently, these food-borne illnesses are only a small portion of the overall ‘burden’ of antibiotic-resistant disease.

A recent study in the Lancet estimated that there are 1.27 million deaths from AMR infections each year. Less than 1% can potentially be directly linked to food-borne illness.19The study, Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis, examined 22 bacteria. Linked to food-borne illness: non-typhoidal salmonella (5620 deaths). Partially linked: enterococcus faecalis (30200 deaths), enterococcus faecium (53500 deaths), escherichia coli (219000 deaths). Not linked: Acinetobacter baumannii Citrobacter spp, (ESBL)-Enterobacter spp, Other enterococci, Group A Steptococcus, Haemophilus influenzae, Kiebsiella pneumoniae, Morganella spp, Mycobacterium tuberculosis, Proteus spp, Pseudomanas aeruginosa, S Paratyphi, S Typhi, Serratia spp, Shigella spp, Staphylococcus aureus, Streptococcus pneumonia (961,680 deaths)
‘Partially linked’ are infections that can come from a variety of sources, with animal-sourced foods being just one of them.
The U.S Centers for Disease Control has identified the 18 AMR ‘threats’, which are “the most alarming antibiotic-resistant germs.”20https://www.cdc.gov/drugresistance/biggest-threats.html
Only 2 of these have a potential direct link to livestock.


Protecting People Requires a One Health approach
Focusing only on animals does not reduce human AMR
Antibiotic resistance is a One Health issue, which means coordinated action is necessary across humans, animals and environment in order to make a difference.

Study from University of Edinburgh explored the relationship between animal use and human AMR, finding that Study from University of Edinburgh explored the relationship between animal use and human AMR, finding that “curtailing the volume of antibiotics consumed by food animals has, as a stand-alone measure, little impact on the level of resistance in humans”.21https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5414261/
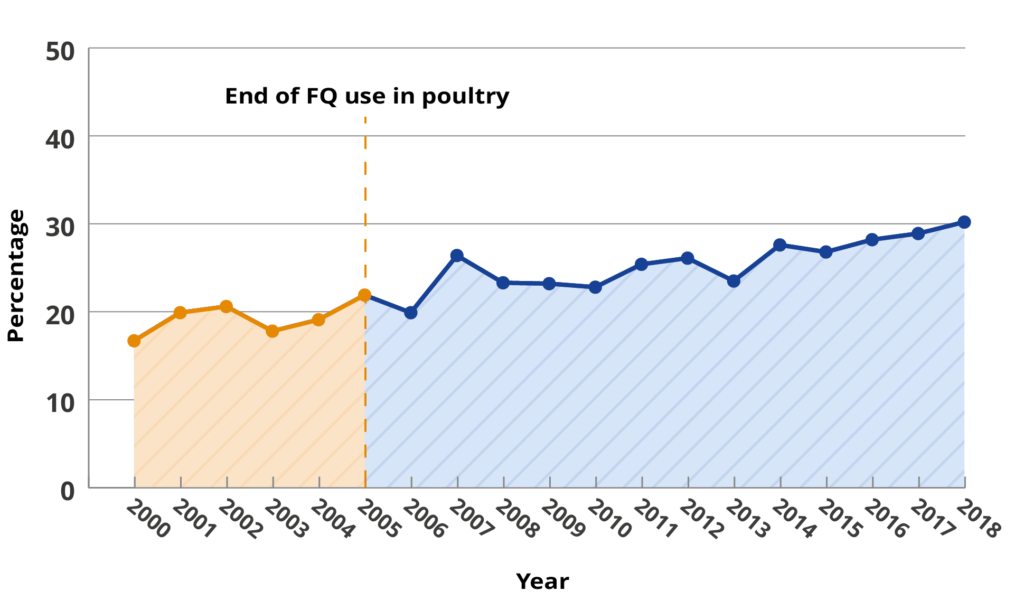
CASE STUDY: Fluoroquinolones in the United States
In 2005, the U.S. halted the use of fluoroquinolones (FQ) in poultry and any extra-label usage in other food animals due to potential that use of these medicines was contributing to rising FQ-resistant campylobacter infections in people.22https://www.fda.gov/animal-veterinary/recalls-withdrawals/withdrawal-enrofloxacin-poultry
However, US government data shows that the rate of FQ-resistant infections in people continued to steadily rise even after use of these medicines was halted in poultry, from just under 20% in 2004 to over 30% today.
This suggests that taking action only in animal use has little effect on human AMR levels, as the University of Edinburgh study found.
Human fluoroquinolone-Resistant Campylobacter Infections in the U.S. (% of total infections)23Data is from the United States’ National Antimicrobial Resistance Monitoring System (NARMS) – https://wwwn.cdc.gov/narmsnow/
Focus on transfer pathways to reduce resistant infections in people
Most antibiotic-resistant bacteria are transferred to people through three primary pathways – from other people, animals and environment. Reducing human AMR requires considering interventions that target where transfer occurs in order to prevent infections and break the cycle of disease.
The European Centre for Disease Control estimates that:
“75% of disease linked to resistant bacteria is due to
healthcare-associated infections”24https://www.ecdc.europa.eu/en/news-events/ecdc-calls-continued-action-address-antimicrobial-resistance-healthcare-settings

The Organisation for Economic Co-operation and Development (OECD) estimates that many of these human infections can be prevented simply by “spending just USD 2 per person a year on measures as simple as handwashing and more prudent prescription of antibiotics.”25https://www.oecd.org/health/stopping-antimicrobial-resistance-would-cost-just-usd-2-per-person-a-year.htm
Other pathways include areas such as environment, travel, and animals. Addressing animal-related AMR bacteria transfer requires measures such as surveillance to track resistance levels / transfer, robust sanitary measures for farm workers, and increased disease prevention to avoid unnecessary use.
“It is now crucial to further address human health”
European Commission on ‘stepping up EU actions to combat antimicrobial resistance in a One Health approach’:26https://ec.europa.eu/commission/presscorner/detail/en/qanda_23_1845

“While much progress has been achieved in the veterinary sector… it is now crucial to further address human health…and increase action in the environmental domain”

“It is also necessary to set recommended targets for AMR and antimicrobial consumption in relation to human health in order to achieve common goals”
Greater capacity for prevention needed in animal health
Animal disease prevention is the primary way to reduce the need for antibiotics and ensure that risk of AMR transfer remains low. This is particularly important in emerging markets where production will grow in the coming decades. However, veterinary capacity and investment in animal health remains low in these markets compared to developed regions. For instance, there are five to six times as many veterinary professionals per livestock animal in Europe compared to Africa.
Ratio of veterinary professionals per livestock animal in Europe versus Africa27WOAH data states the ratio of veterinary professionals per livestock unit is 3530:1 in Africa and 612:1 in Europe. Source: WOAH 2022 Observatory Annual Report


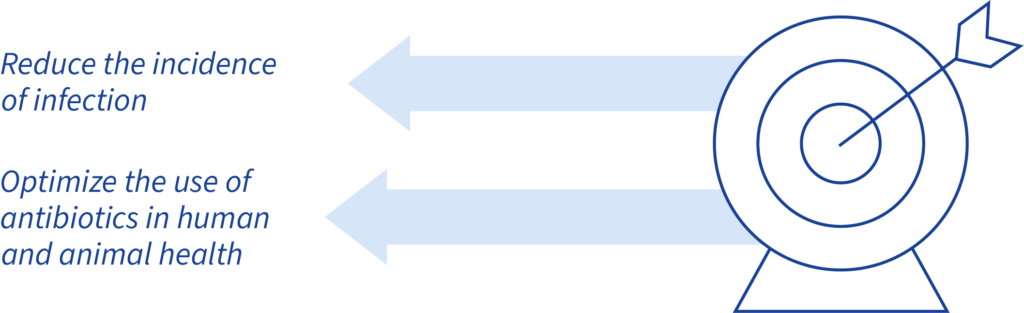
Goals need to be focused on prevention and optimized use
194 countries across the world signed on to the Global Action Plan on AMR in 2015, which accelerated efforts to tackle this challenge. At the core are two key objectives that address disease and antibiotic use. This approach recognizes that prevention is essential, and efforts must focus on optimizing use, not simply reducing the use of antibiotics. This recognizes that antibiotics will always be necessary for treating bacterial disease and supporting animal welfare.
Objectives of the World Health Organization’s Global Action Plan on AMR28https://www.who.int/publications/i/item/9789241509763
- 1
- 2
- 3Data provided by CEESA. Their International Sales Survey covers sales from the 9 largest global animal health manufacturers (all HealthforAnimals Members) and local/regional manufacturers in Italy, Spain, the UK and Latin America.
- 4Source: Antimicrobial Resistance in the EU/EEA: A One Health Response, https://www.ecdc.europa.eu/sites/default/files/documents/antimicrobial-resistance-policy-brief-2022.pdf
- 5Source: Antimicrobial Resistance in the EU/EEA: A One Health Response, https://www.ecdc.europa.eu/sites/default/files/documents/antimicrobial-resistance-policy-brief-2022.pdf
- 6
- 7Calculation reflects the forthcoming WHO Medically Important Antimicrobial List where all penicillins are now a ‘highly important’ antimicrobial. Globally: WOAH, 7th Annual Report on Antimicrobial Agents Intended for Use in Animals, USA: USFDA, 2021 Summary Report On Antimicrobials Sold or Distributed for Use in Food-Producing Animals, Europe: 2023 ESVAC Report
- 8Calculation reflects the forthcoming WHO Medically Important Antimicrobial List where Macrolides are no longer an HP-CIA. WOAH’s ‘7th Annual Report on Antimicrobial Agents Intended for Use in Animals’ states that fluoroquinolones and 3rd & 4th gen cephalosporins are 3.4% and 0.6% of animal use respectively. WOAH also stated in direct correspondence that bacitracin is 90% of polypeptide use (8.7%), meaning that only 10% (.87%) could potentially be polymixins, an HP-CIA.
- 938.6% of human use is Watch antibiotics. Reserve is less than 1%. https://www.cidrap.umn.edu/antimicrobial-stewardship/global-use-broad-spectrum-antibiotics-rising-data-show
- 10
- 11
- 12
- 13List of Antimicrobial classes authorized for humans and animals on page 24: https://cdn.who.int/media/docs/default-source/antimicrobial-resistance/amr-gcp-irc/who_mialist_draft_forexternaldiscussion.pdf?sfvrsn=af6f2ebf_1. Percentages of use in animals can be found in the WOAH Seventh Annual Report on Antimicrobial Agents Intended for Use in Animals
- 14
- 15
- 16
- 17
- 18
- 19The study, Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis, examined 22 bacteria. Linked to food-borne illness: non-typhoidal salmonella (5620 deaths). Partially linked: enterococcus faecalis (30200 deaths), enterococcus faecium (53500 deaths), escherichia coli (219000 deaths). Not linked: Acinetobacter baumannii Citrobacter spp, (ESBL)-Enterobacter spp, Other enterococci, Group A Steptococcus, Haemophilus influenzae, Kiebsiella pneumoniae, Morganella spp, Mycobacterium tuberculosis, Proteus spp, Pseudomanas aeruginosa, S Paratyphi, S Typhi, Serratia spp, Shigella spp, Staphylococcus aureus, Streptococcus pneumonia (961,680 deaths)
- 20
- 21
- 22
- 23Data is from the United States’ National Antimicrobial Resistance Monitoring System (NARMS) – https://wwwn.cdc.gov/narmsnow/
- 24
- 25
- 26
- 27WOAH data states the ratio of veterinary professionals per livestock unit is 3530:1 in Africa and 612:1 in Europe. Source: WOAH 2022 Observatory Annual Report
- 28
